One of the things Sheila Irvine missed most after she lost her sight was being able to read.
“Reading, that’s in my DNA,” says the 70-year-old, who lives in the small English town of Malmesbury, in the Cotswolds. “I am an avid bookworm. As a child I read a paperback every week.”
So when she heard about a clinical trial to insert a microchip in the back of her eye that could potentially help her see letters and words again, she signed up. “I was nervous, excited, all those things,” she says.
Irvine began noticing vision problems in her 30s and eventually lost her sight, slowly, from dry age-related macular degeneration (AMD), a disease that affects 1 in 227 people worldwide and is the most common form of incurable blindness in older adults. “At first, I noticed I couldn’t judge things when I was driving,” she says. “I kept hitting the pavement. I cried all day long when I sent my driving license back over 15 years ago, but then I said to myself, ‘Move on and get on with things.’ ” She values her independence and visits with friends at her local pub regularly, she says.
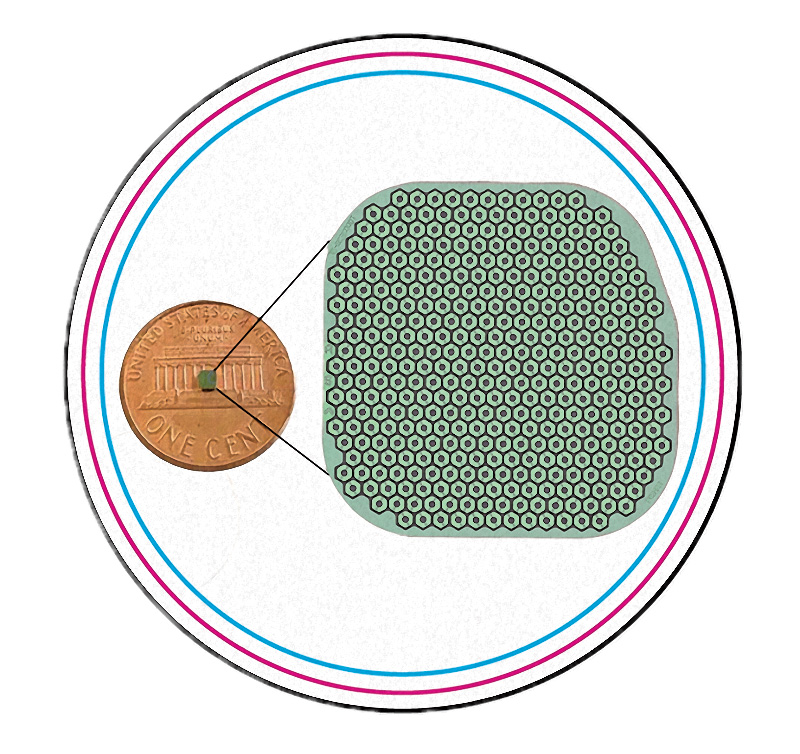 Illustration: Science Corporation
Illustration: Science Corporation
To participate in the clinical trial, Irvine took a two-hour journey on public transit to Moorfields Eye Hospital in London, where she was one of five patients implanted with the microchip, a device invented by Stanford ophthalmology professor Daniel Palanker. During the two-hour procedure, surgeon Mahi Muqit inserted the microchip—shaped like a tiny SIM card—under the center of her retina, a spot called the macula. The trial included a total of 38 people across five European countries with geographic atrophy, an advanced stage of dry AMD that causes the death of nerve cells in the macula, creating blind spots for reading, driving, and recognizing faces.
Of the 32 patients who stayed in the trial for a year, 27 of them regained the ability to read letters, numbers, and words using a special pair of high-tech glasses. The results were published in the New England Journal of Medicine in October 2025. It was the first time a prosthesis restored functional vision to patients with incurable vision loss, giving them the ability to see shapes and patterns rather than just light, as past prosthetics have. With digital enhancements enabled by the glasses such as zoom and contrast, some patients were able to achieve the equivalent of 20/42 vision after one year, and the researchers plan to continue enhancing the system’s capabilities.
Irvine says the two-hour surgery, which was painless, has been transformative. “It was dead exciting when I began seeing a letter. Now I read most days. I read so much. I’m fascinated with Jack the Ripper and read all those books,” she says during a recent Zoom interview that includes her London surgeon, Muqit, as well as Palanker, calling in from California. During the interview, she’s introduced to Palanker as the inventor of the microchip—called PRIMA, for photovoltaic retina implant microarray—and her eyes widen.
“My darling man,” she says, addressing Palanker. “Thank you so much for what you’ve done for me. You can imagine how I feel. It’s changed so much.”
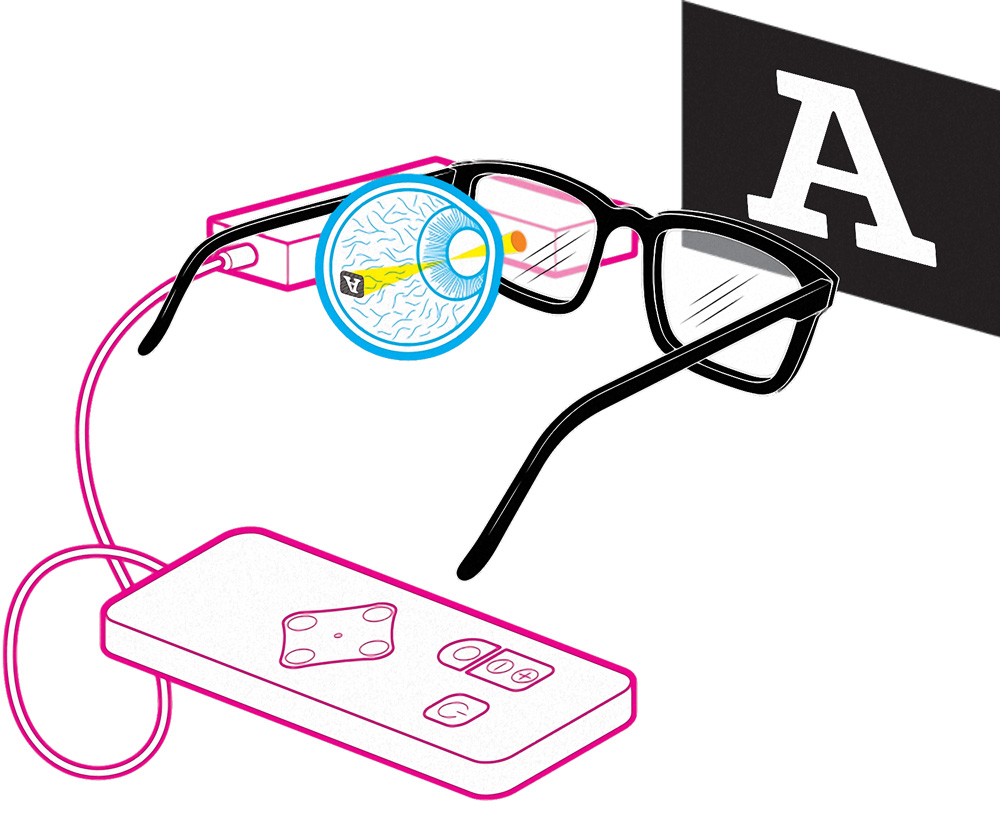 DECODER: The retinal implant converts infrared light into electrical current that stimulates the brain. A pocket controller enables adjustments such as magnification and contrast. Illustration: McKibillo
DECODER: The retinal implant converts infrared light into electrical current that stimulates the brain. A pocket controller enables adjustments such as magnification and contrast. Illustration: McKibillo
Palanker first envisioned such a device 20 years ago, when he was working with ophthalmic lasers used to treat eye conditions. He figured that since the biological vision system is transparent, he could build a chip that delivered information using natural light. The system would use glasses similar to virtual reality devices, he wrote in a 2005 paper.
Dry macular degeneration causes slow deterioration and death of the photoreceptor cells at the macula, that central part of the retina that is densely populated with them. Photoreceptors are specialized cells that convert light into neural signals that get sent to the brain, enabling vision. People with the disease lose their central field of vision. (Irvine describes this as seeing two black discs in the middle.) “With dry macular degeneration, the photoreceptors just start dying,” Palanker says. “This causes irreversible damage. You lose vision. The whole area where they are dead is a blind spot.” Most patients do, however, maintain some peripheral vision by retaining other retinal cells. And therein lay Palanker’s opportunity.
‘I met an architect recently in Rome who is using it for work, which is remarkable. He wants to complete the design of a church.’
Palanker proposed a plan to help restore some of the lost central vision back in these patients. The idea was to build a wireless retinal microchip that would work as a prosthesis to replace the lost photoreceptors. It would rely on infrared light, which would be invisible to the photoreceptors in the periphery, so that patients could harness both their natural peripheral vision and prosthetic central vision.
Palanker designed the chip to convert light into electrical current that stimulates inner retinal neurons to communicate with the brain. It was first manufactured at Stanford’s Nanofabrication Facility for testing and is now made by Science Corporation. The chip is made up of 378 light-powered pixels that use photovoltaic technology to convert light into electricity. “It’s basically like solar panels,” Palanker says. “Every pixel is a little solar panel in the eye.”
The microchip works in tandem with augmented-reality glasses that have a small video camera mounted in front of one eye. The camera captures images and converts them into patterns of infrared light that get transmitted to the retinal implant. A month after surgery, patients begin a training process that typically takes several months, learning how to interpret the new light signals that are seen by them as visual patterns in black and white. Objects such as letters are placed in front of them while they practice positioning the glasses correctly and using the adjustments on a pocket controller.
 Daniel Palanker (Photo: Steve Fisch)
Daniel Palanker (Photo: Steve Fisch)
“At first I thought, ‘I’m never going to get this,’ ” Irvine says. “You have to keep your head steady. That took some doing. You have to be patient with yourself.”
Palanker says the process is similar to learning how to use a cochlear implant, which requires routine practice to teach the brain how to recognize prosthetic visual input. He wasn’t sure how user-friendly it would be. But according to the study, most recipients saw letters right away and were using PRIMA at home to read within a few months of implantation.
“The fact that they use it daily to read, to play games and do things like crossword puzzles and cooking, that’s all very exciting,” he says. “I met an architect recently in Rome who is using it for work, which is remarkable. He wants to complete the design of a church that, because of his vision impairments, he couldn’t do until now.”
PRIMA is the culmination of decades of development, prototypes, animal trials, and a small first-in-human trial. Frank Brodie, a former fellow in Palanker’s lab who is now Science Corporation’s medical director for vision and PRIMA, says the company is expecting to get European approval to make the device available to the general public by summer and is working to get approval from the Food and Drug Administration for use in the United States.
Meanwhile, Palanker and his lab continue working on new versions of the microchip, increasing the number of pixels for better resolution, and designing new software for possible facial recognition.
‘It’s basically like solar panels. Every pixel is a little solar panel in the eye.’
“Currently what we use in clinical trial is binary, like black and white, and that’s not great for faces,” he says. The next-generation chip may boost pixel density above 10,000, offering an increase in image quality, and the software will be adjusted to better represent faces in hopes that patients will be able to recognize other people. Color recognition may be possible further down the line, he says, adding that the system was designed to accommodate upgrades.
Irvine, for one, is excited about the potential for a new version of PRIMA. “People are still just silhouettes,” she says. “I can’t see faces. I’ll try it!”
Tracie White is a senior writer at Stanford. Email her at traciew@stanford.edu.



